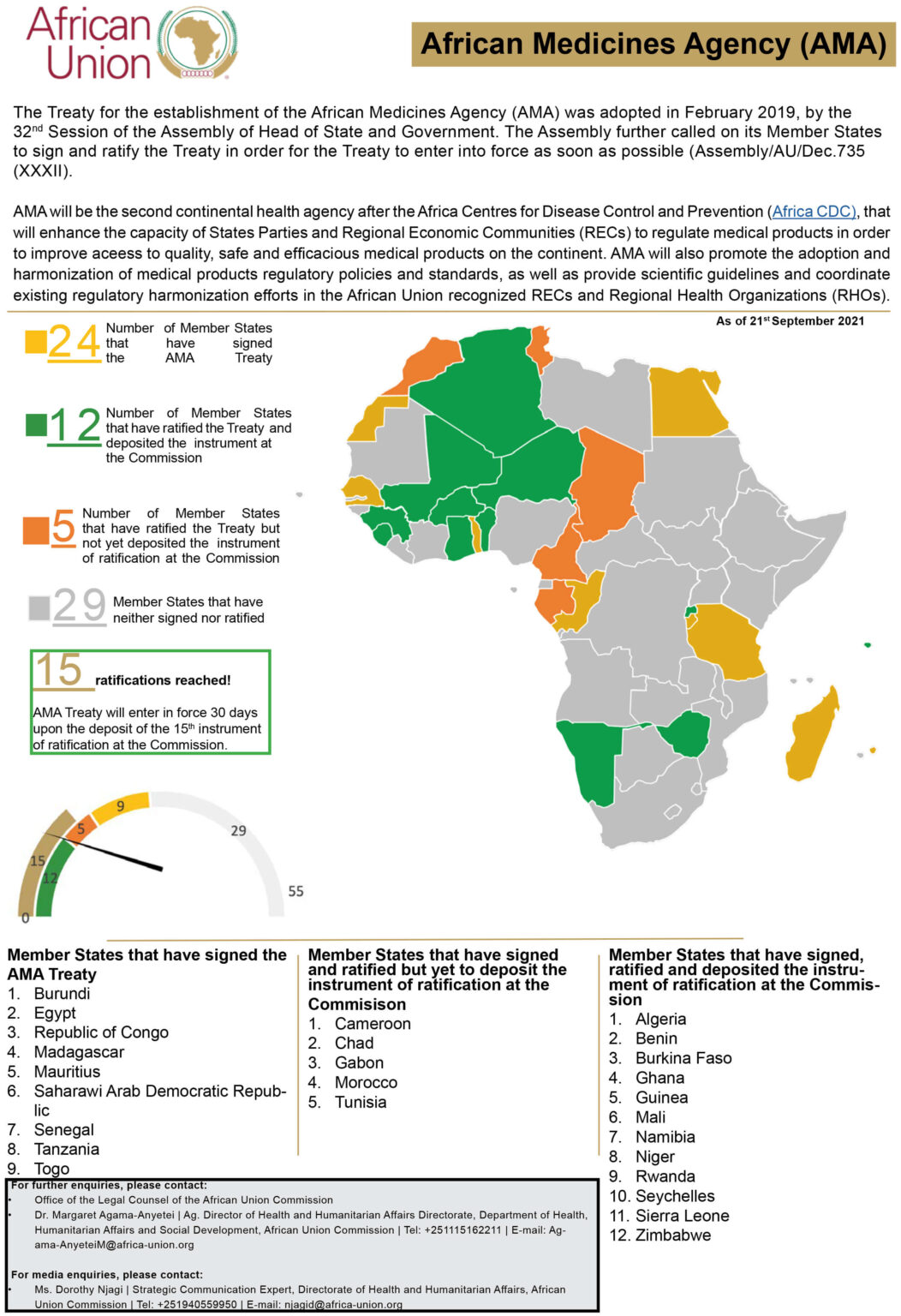
Patients in Africa are a step closer to speedier access to newer, safer medicines following last week’s ratification of the African Medicines Agency (AMA) treaty by 15 African Union member countries.
On 5 October 2021, Cameroon became the 15th country to formally notify the African Union Commission that they had ratified the treaty, finally pushing the AMA over the threshold into reality. A further three countries have ratified the treaty but not formally informed the AU, while eight more have signed the treaty but not yet ratified it. This means 36 of Africa’s 55 countries have expressed support for the AMA. However, African powerhouses Ethiopia, Nigeria, and South Africa have not yet expressed their support for the agency.
The AMA represents, for many countries, the promise of the development of local production, and the development, across the continent, of centers of excellence for research, the strengthening and security of supply chains, for the maintenance of a healthy environment and the fight against falsified medicines.
The full implementation of the African Medicines Agency will be a game-changer for all Africans to access safe, effective, and quality medical products in a timely manner.
Greg Perry, Assistant Director-General, The International Federation of Pharmaceutical Manufacturers & Associations
Originally published on Health Policy Watch.